A Primer on Performance Plastics
from good, to better, to best
Editor’s note: To download this article as a PDF, please click here.
I just returned from the annual conference of the International Association of Plastics Distribution (IAPD), where I gave a presentation on the use of performance plastics. As the name suggests, IAPD is an organization dedicated to supporting the needs of suppliers, distributors, fabricators, and other service providers involved in the sales and distribution of plastic materials. It was a great conference – with lots of interaction.
IAPD is an organization that I am still getting familiar with – but I also have to say that it is one of the hidden gems of the plastics industry. As an example, I learned more about fluoropolymers during the four day conference than I had learned in my previous 30 years in the plastics industry (including the ten years I spent as Application Engineer for DuPont – the company that invented Teflon).
What Are Performance Plastics?
To some people, the phrase Performance Plastics may seem like an oxymoron: a figure of speech in which seemingly contradictory terms appear side by side, in the same manner as the phrases awfully good, and genuine imitation. We all know that plastics are cheap, flimsy, practically worthless. Furthermore, the word plastic is often synonymous with fake – plastic flowers, plastic people, plastic surgery. So how can a cheap, flimsy, practically worthless material – one that is basically fake – be considered a performance material?
The answer is twofold: first, performance (like beauty) is often in the eye of the beholder, and second, the plastics we are talking about are not the cheap, flimsy materials that your grandmother used to complain about – these are Performance Plastics, and they are bona-fide, certified, and fortified, and can often wreak havoc on the performance of traditional materials such as tin, iron, copper, and steel. And while they may not be cheap, there is an old adage – you usually get what you pay for.

But what exactly is performance? I prefer to think of performance as something that can be measured in specific and absolute terms, using commonly accepted means of measurement. We are not using arbitrary units of measurement (such as a digit – a unit of length based on the width of a finger, or a stone – a unit of weight based on the mass of a rock of a certain size). Instead we are using standardized units of mass, length, and time. And when we measure the performance of these materials, we find they have repeatable and predictable properties. We often organize the data we obtain on these properties into different categories as described below. While the manner in which we organize the data may vary, the important thing to remember is that the measurement data is repeatable and predictable, and we can rely on this data to predict the performance of the material in the application where are using it.
Properties a Mechanical Engineer Can Relate To
Mechanical Properties
Mechanics is the area of science that deals with the behavior of physical bodies as they respond to force or movement. Along with materials science, it is one of the disciplines that mechanical engineers use in their design of physical systems. Taken together, mechanical engineers rely on the mechanical properties of materials, including the basics of strength, stiffness, and toughness. We want to understand how the materials we use respond under tension, compression, flexure, torsion, and shear. We want to know how much they elongate under a given load, and whether the deformation is temporary or permanent. We want to know how hard – or soft – a material is, and how it responds to impact and abuse.
Chemical Resistance
In addition to mechanical properties, we want to know how the material is affected by exposure to chemicals, regardless of whether these chemicals are nice – or downright nasty. This includes acids, bases, detergents, cleaning agents, oils, greases, solvents (polar and non-polar), and combinations of the above. Most of the time, the material will only be exposed to the chemical for a short duration, but there are times when the the exposure is constant and long term. Can the material still perform?
Electrical Properties
Sometimes, mechanical engineers also want to know how a material responds when it is subjected to an electrical charge, or is in the presence of an electric current. While we often think of plastic materials as being non-conductive electrically (in other words they are good insulators), there are many variables to consider in the design of an electrical system. What is the electrical charge? What is the current flow? What are the thicknesses of the various components in the system?
Temperature Extremes
Then there are temperature extremes – both low and high. At the low end, when things are really, really cold, materials often get brittle – not just plastics, but all kinds of materials. What happens when a given plastic is exposed to extreme cold temperature for a few days? Or a few weeks? Or several years?
At the opposite end, there is high heat exposure. What happens to the material when it is exposed to 100 °F for a few days? Or 200 °F for a few hours? Or 300 °F for several weeks? Or can we go to even higher temperatures? Do you have the data to confirm that?
Other Extreme Environments
Finally, there is performance in severe environments, including exposure to sun shine and UV light, as well as gamma rays, x-rays, and other forms of radiation.
The Bottom Line
Performance plastics are plastic materials which have repeatable and predictable properties under a broad range of end-use environments, including exposure to chemicals and extreme temperatures.
Polymer Chemistry for the Mechanical Engineer
All plastic materials are polymers, meaning they are made of molecules consisting of many parts. (The word polymer comes from the Greek word polumeres – polus, meaning many, + meros, meaning part or component). Plastic molecules typically consist of atoms and small compounds of carbon, hydrogen, nitrogen, etc. The atoms and compounds are first bonded together into a small molecule, called a monomer. These monomers are then bonded to one another to form a chain in what is known as a polymerization process. This chain is a polymer molecule.
Most of the plastics we are familiar with are based on hydrocarbon chemistry (organic compounds containing hydrogen and carbon atoms), although other chemistries may come into play. The polymerization process can be simple or complex, depending on the monomers involved, and the final polymer. I often think of the process as The Marriage of the Mers. For the mechanical engineer, what is of more importance is the de-polymerization process, or The Divorce of the Mers. How a given depolymerizes – and what variables are involved – often dictates the performance in the end-use environment.
(Molecular) Size Matters
Polymer molecules, even molecules of the same chemistry and type, come in different sizes. The size can be measured by the overall chain length, or in the mass (or weight) of the molecule. A longer chain length not only results in a higher molecular weight, it also results in better physical properties (higher strength, higher stiffness, better impact resistance, etc.). For any given resin, there will be a mix of lengths in the molecules in that material, and a mix of molecular weights. This mix can be measured and quantified in a number of ways, including the average molecular weight (AMW) and the molecular weight distribution (MWD).
Why should you care about this?
Molecular weight not only influences physical properties, it also affects the characteristics of the polymer in a number of areas – including how it performs in its molten state (how it melts, how it flows, how it cools and shrinks and solidifies). In other words, the molecular weight is a key factor in how a given polymer can be processed. Can it be cast? Injection molded? Extruded? How does it respond to machining operations? What are the limitations of processing that polymer using that process?
Furthermore, we need to remember that all polymer materials go through a number of processing cycles. Even a prime virgin resin intended for use in injection molding has been through at least one melt processing cycle. If the material has been processed into a stock shape (via casting, or extrusion), it has probably gone through two melt cycles. And if it has been modified or compounded it may have gone through several. Each time a polymer goes through a melt cycle, it affects the chain lengths of the polymer molecules.
One of the responsibilities of a material supplier is to ensure that the material has an AMW and MWD within a certain range, and that its physical and mechanical properties meet certain specifications. Most material buyers have a protocol in place to verify compliance with these specifications as well. The important thing to remember, molecular weight is a critical parameter – not just for end-use performance but for processing as well.
Meet the Family
Polymers are often categorized into different types, such as thermoset and thermoplastic, or amorphous and semi-crystalline. While this categorization can be helpful, another method of classification is to describe the chemical family of the resin, specifically the base monomer. This is a primary means of classification within the plastics industry, since the chemistry is inter-related to the feedstocks and catalysts, the chemical intermediates, and the method(s) of polymerization. However, it also plays an important role in material selection, as the chemistry of the polymer plays an important role in the properties of the material.
A given chemical family may have many related polymers. One example is the nylon family, better known as polyamides (PA). There is PA 6, PA 6/6, PA 11, PA 12, PA 612, PA 1012, etc, with the numbers designating different variations of monomers and the amide bonds between them. And while each type of nylon has slightly different properties, as a family, they behave in a similar fashion to each other.
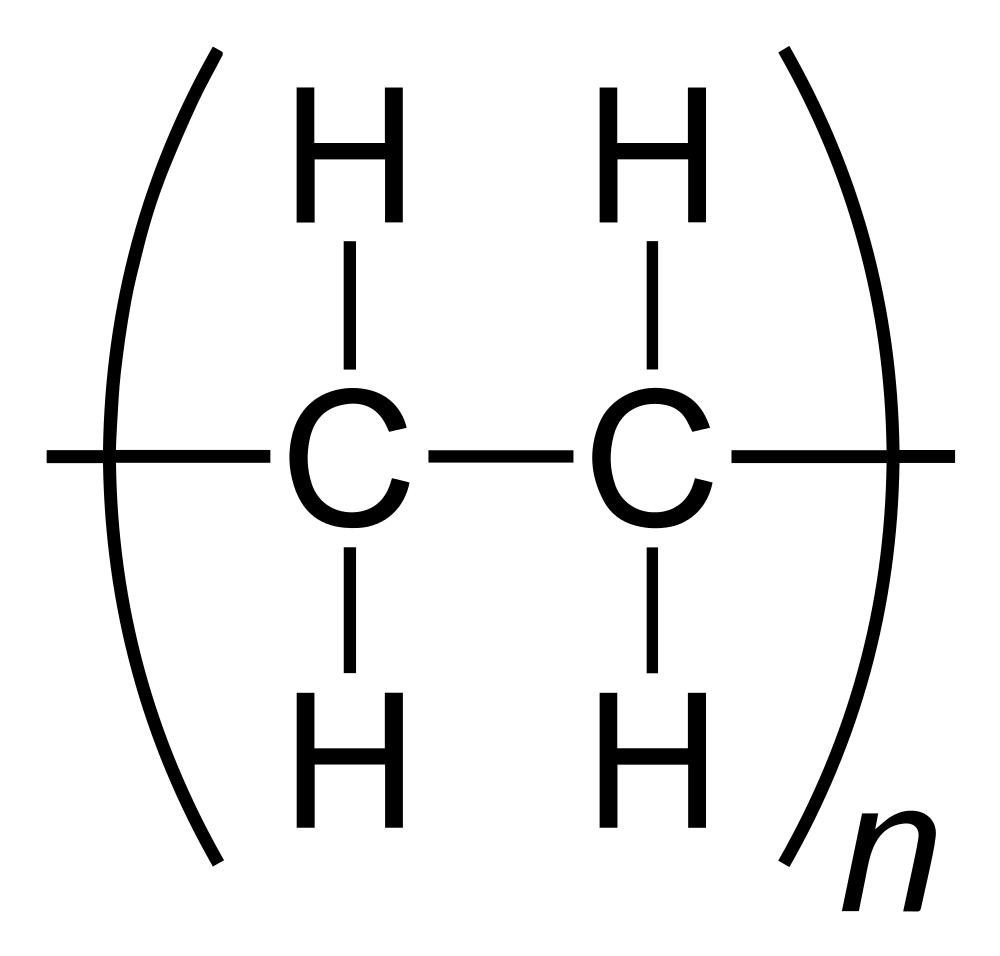
Another example is the polyethylene (PE) family, with low density PE, high density PE, linear low density PE, etc. Again, while while each type of polyethylene has slightly different properties, as a family, they behave in a similar fashion to each other.
Traditional Polymers
While most people think of plastics as a synthetic (man-made) material, there are many naturally occurring polymers. But natural or synthetic, most polymers consist of a chain of monomers, bonded together via various combination of hydrogen-carbon bonds. The polymer chain may be linear, or it may be branched, and occasionally even cross linked (bonds between different polymer chains). The performance of any given material will depend on the chemistry of the chains, and the molecular weight.
Aromatic Polymers
Aromatic polymers usually provide a significantly higher level of performance than traditional polymers. In organic chemistry, the term aromatic refers not to the way something smells, but to the way molecules bond in a ring form, creating strong and stable molecular bonds that are often superior to molecular bonds made in a linear fashion. In polymer chemistry, aromatic polymers have aromatic ring bonds in the molecule, and as a result the material typically has performance advantages over a traditional polymer.
As an example, you may be familiar with the term aramid, which is used to describe an aromatic polyamide. (Kelar is an aramid fiber, Nomex is an aramid available in both fiber and sheet form). One way to categorize an aramid is to simply think of it as nylon on steroids.
I often think of aromatic polymers as polymers on steroids.
Fluorinated Polymers
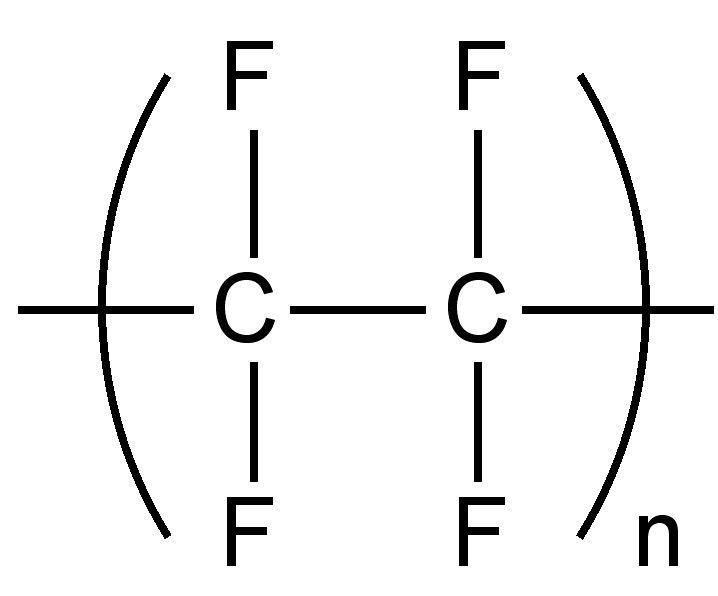
Another step improvement in performance is via the use of a fluorinated polymer (fluoropolymer). Instead of the hyrdogen-carbon bonds in traditional and aromatic polymers, fluoropolymers have strong carbon-fluorine bonds. Fluoropolymer typically have incredibly high resistance to solvents, acids, and bases. While there are many chemical families involved, an easy way to categorize them is to look at the chemical family.
Going back to our example of polyethylene (PE), you may note that it is made from the ethylene mononer, with the chemical formula, C2H4, meaning it consists of two carbon atoms, and 4 hydrogen atoms.
If we replace each hydrogen atom in the monomer with a fluorine molecule we have the tetrafluoroethylene monomer, C2F4, meaning it consists of two carbon atoms, and 4 fluorine atoms (tetra meaning four). If we polymerize this monomer, we get the polymer polytetrafluoroethylene (PTFE). PTFE is best known by the brand name Teflon. It has extraordinary properties, not just in chemical resistance, but in resistant to heat, and ultra low friction (the classic non-stick material on frying pans).
I know why they call them fluoropolymers, but the name can be confusing,
because their performance is through the roof.
If you learn a little bit about the chemical family, and the chemistry involved in the family, it can help you can understand the behavior of a given polymer. It’s like having the secret decoder ring to the world of polymer chemistry.
A Little Bit About Brands
As a consulting engineer, I am frequently asked to make recommendations on the selection – and specification – of a suitable material for a given application. However, as an independent consulting engineer, I am often reluctant to recommend one brand over another.
I understand the importance of branding in the material supply industry. It is one way to ensure the quality and consistency of a material through the entire supply chain. I am also a firm believer that product quality is best achieved through proper control of the materials used in the manufacturing process – and the consistency of those materials is a critical parameter.
So, while I may be reluctant to recommend one brand over another, I do want to make a distinction between specifying a given brand of material, and allowing for the use of a generic material.
When you specify the use of a certain material from a certain resin supplier, you are basically saying:
We have evaluated the use of this material in our application, and we are confident that it meets our needs. We are also confident that this supplier will provide this material in a manner that meets or exceeds international standards for resin consistency.
And, while you may not realize it, you are also saying:
We are confident that all of the partners in the material supply chain adhere to the international standards and code of ethics set by this brand.
When you allow the use of a generic material, you are basically saying:
We have evaluated the use of this material in our application, and we are confident that it meets our needs. If the supplier can provide a material that meets the industry definition for this material, we’ll use it.
As you can imagine, this can result in a wide variation in the material that is actually being used in the fabricated parts, depending on the purity and consistency of the source material. You might also want to add another note, saying something to the effect of:
Supplier to verify the consistency of the material meets international standards for resin consistency.
Key question: can you rely on your material provider to ensure all that?
Using Performance Plastics
I have been building things for as long as I can remember. It started when I was a kid with a set of Tinker Toys, followed by Lincoln Logs and Lego blocks and Erector Sets. When I got older, I started building tree houses and forts, then model cars and model airplanes, toothpick bridges, you name it. I loved exploring how things went together, and how they came apart. Sometimes I would repair something that had been broken, and then present it proudly to my parents (or my grand parents), only to have them look me in the eye and ask, Are you sure this is going to work?
Years later, I went to engineering school, and got my degree in engineering, and then my certification as a professional engineer. Today I work as a consulting engineer, helping companies design and develop new products, most of which involve the use of plastics. In the process, one of the things I have learned:
The best design in the world is worthless if you are using the wrong material
(or if you are using a substandard material)
Although, truth be told, I don’t know which is worse:
A. Using a material that 100% certified
(but is wrong for this application)
or
B. Using a material that is sub-standard and unpredictable
(but just seems to work in this application, some how, some way)
The bottom line, performance plastics are not for everyone. If you just want to use SKP (some kinda’ plastic) in your product, then go right ahead. But if you want to design and manufacture a product using a performance plastic, I recommend you explore the following questions before you proceed:
- Do you need a traditional performance polymer, or an aromatic polymer, or a fluorinated polymer?
- Do you want to specify the exact brand and material code? Or do you want to allow the use of a generic material?
- Can your material supplier verify the average molecular weight (AMW) and the molecular weight distribution (MWD) of the material they supply?
- What additives are in the resin, and what is the effect of a melt cycle on those additives?
- What is the manufacturing process you are using?
- What effect does the manufacturing process have on the material you selected?
- What effect does the material you selected have on the manufacturing process?
- What are you going to tell your grandmother when she looks you straight in the eye and asks you, Are you sure this is going to work?
Once you have the answers to the above questions, you are on your way to the successful use of a performance plastic.







Want to hear more from plastics guy?
Sign up for a monthly update of our latest posts.
You have Successfully Subscribed!